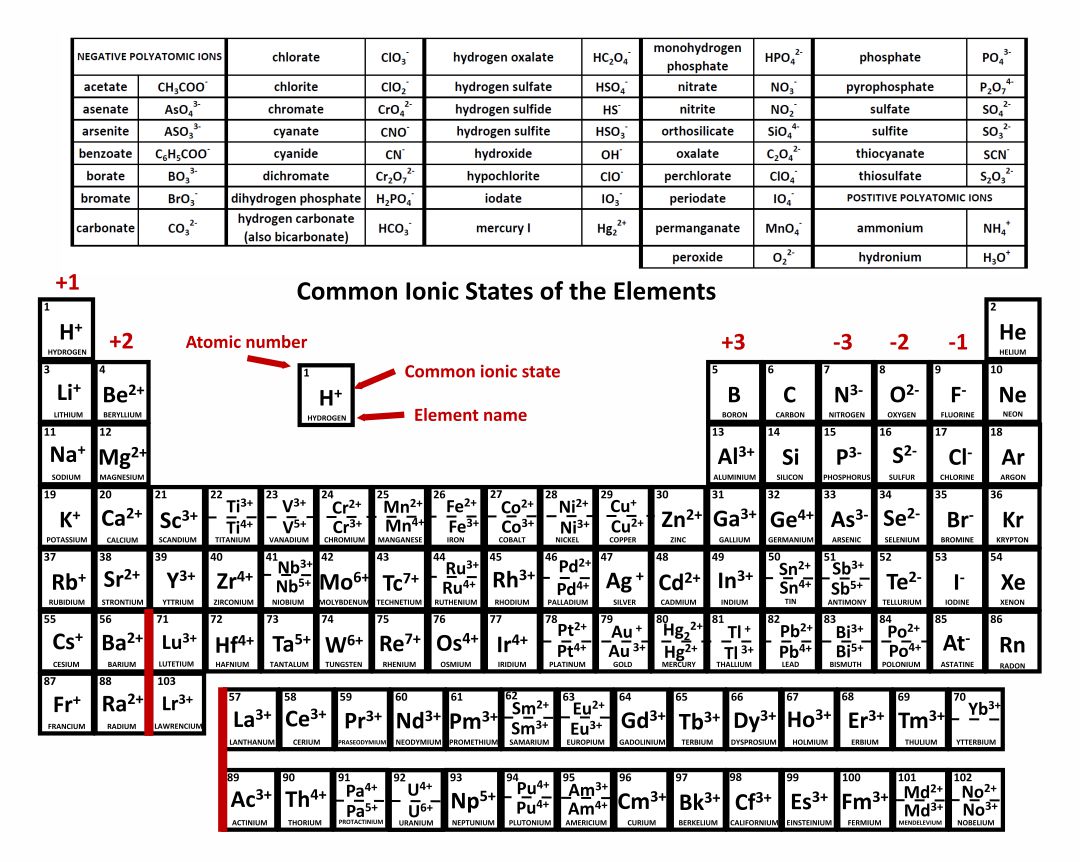 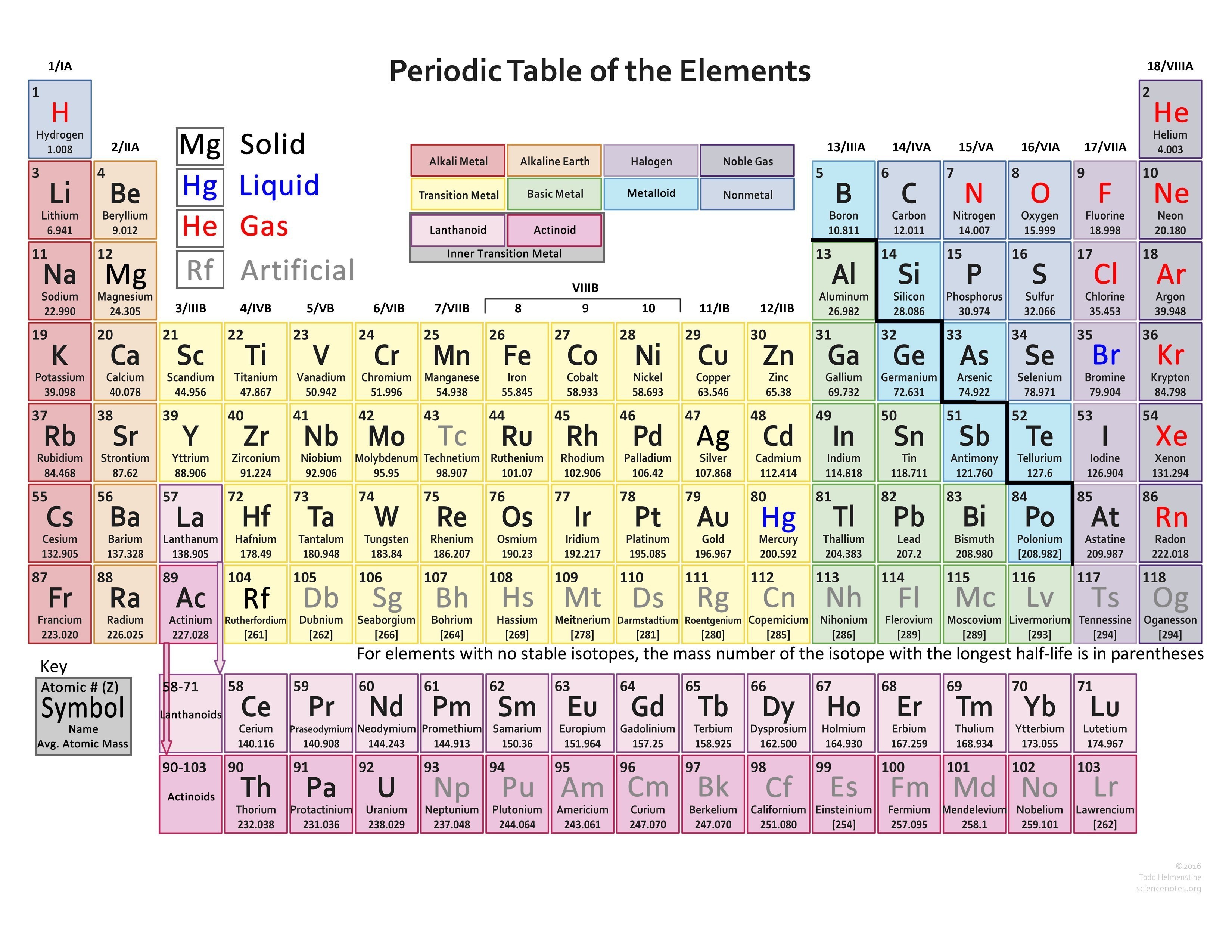
It consists of 7 lines (horizontal), which are also called periods.The element table is divided into period groups: main and sub-groups (vertical), a total of 18 groups.Mass and atomic radius increase from top left to bottom and right (with some exceptions).Up to 118 protons in the nucleus of the element Ununoctium are currently known. The first element with the atomic number 1 is hydrogen. This indicates how many positively charged particles (protons) are present in the atomic nucleus. The elements are arranged according to their atomic number.The well-known PSE refers to the atoms in their electrically neutral state, which means that the same number of negative electrons (-) are present in the electron shell as protons (+) in the atomic nucleus.The Mendeleev table of elements has prevailed to this day.ġ23Movies Periodic table: explanation of the elements But who invented the periodic table of the elements? As early as 1869, the Russian Dmitri Mendeleev and the German Lothar Meyer came up with a similar idea to represent the chemical elements. The chemistry periodic table appears confusing at first glance, but the information in the element table has a logical explanation and is less complex once you have understood the PSE. If you want a good WordPress PageBuilder check out divi discount from Elegant Themes. Why is this element table used in chemistry? And how does the periodic table of the elements work? We provide you with the periodic table explanation in our guide, so that you keep an overview of main periodic table groups and atomic numbers and reveal where you can get a periodic table to print out. The chemical elements are shown in a table in the Periodic Table of the Elements (PSE for short) and should not be missing in any table work.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
